SILICON NITRIDE The IDEAL BIOMATERIAL
In the race to achieve interbody fusion, material matters.
What is SILICON NITRIDE (Si3N4)?
Silicon Nitride, a non-metallic ceramic composed of the essential elements silicon and nitrogen, stands out as a superior biomaterial for fusion. Its unique combination of antibacterial, osteoconductive, and osteoinductive properties promotes bone growth and healing. Its hydrophilic nature, strength, and favorable imaging characteristics further distinguish it from traditional PEEK or Titanium implants.

Si3N4 PROPERTIES
Antibacterial
Surface properties actively repel and/or kill various types of bacteria, reducing infection risk.
Bone-Building
Unique surface chemistry mechanisms continuously stimulates new bone growth and integration.
Hydrophilic
Attracts fluids, further deterring bacteria colonization and enhancing bone-building mechanisms.
Imaging-Friendly
Unlike metal implants, allows for clear visualization in X-rays, CT scans, and MRIs.
Superior Benefits
Our Silicon Nitride offers superior conditions for fusion, providing enhanced stability and durability compared to PEEK and Titanium.

Si3N4 in Medical Implants
Silicon nitride (Si3N4) is proving to be a game-changer in spinal fusion surgery. One of the studies published in The Spine Journal found a 80% fusion rate at 3 months post-operatively in patients who received Si3N4 cervical interbody implants, indicating its effectiveness in promoting bone growth (1).
In addition, a separate multicenter study demonstrated a 90.2% fusion success rate and only 3.1% of patients requiring revision surgery due to complications, further supporting Si3N4’s superior performance (2). The material’s radiolucent properties also offer enhanced visualization during surgery and post-operative imaging, contributing to improved surgical precision and patient care (3).
Ball HT, McEntire BJ, Bal BS (2017) Accelerated Cervical Fusion of Silicon Nitride versus PEEK Spacers: A Comparative Clinical Study. J Spine 6: 396. doi:10.4172/2165-7939.1000396 2.Mobbs, Ralph James et al. “Clinical outcomes for lumbar fusion using silicon nitride versus other biomaterials.” International Orthopaedics vol. 45,4 (2021): 925-934. doi:10.1007/s00264-020-04823-2 Anderson, Paul A et al. “Silicon Nitride Ceramic: A New Biomaterial for Spinal Arthrodesis.” The Spine Journal vol. 13,10 (2013): 1209-1217. doi:10.1016/j.spinee.2013.06.010
Percentage of New Bone Around Implant at 3 Months
PEEK
8%
24%
Titanium
19%
36%
SILICON NITRIDE
65%
71%
Implant Interface
Surgical Area
Webster TJ, Patel AA, Rahaman MN, Sonny Bal B. Anti-infective and oste ointegration properties of silicon nitride, polylether ether ketone), and titanium implants [published online ahead of print July 31, 2012]. Acta Biomater.
Key Advancements in Si3N4 Technology
Superior Bone Growth and Healing
The surface chemistry and natural nanostructure topography of silicon nitride provide an optimal environment for stimulation of osteoprogenitor cells to differentiate into osteoblasts.
Sustained Release of Growth Factors
Continuously delivers signaling molecules that stimulate the production of new bone cells.
In Vitro Study May not be representative of clinical performance
Enhanced Bone Formation and Preservation
Promotes the growth of new bone tissue, extends the lifespan of existing bone cells, and prevents bone breakdown.
Wistar Rat Study
Wistar Rat Study May not be representative of clinical performance
Improved Bone Healing
Facilitates the formation of new blood vessels, delivering essential nutrients and oxygen for robust bone repair.
In Vitro Study May not be representative of clinical performance
1. Webster TJ, Patel AA, Rahaman MN, Sonny Bal B. Anti-infective and osteointegration properties of silicon nitride, poly(ether ether ketone), and titanium implants [published online ahead of print July 31, 2012]. Acta Biomater.
2. Gorth DJ, Puckett S, Ercan B, Webster TJ, Rahaman M, Bal BS. Decreased bacteria activity on Si(3)N(4) surfaces compared with PEEK or titanium. Int J Nanomedicine. 2012;7:4829-4840.
Multifaceted Antibacterial Action
Silicon nitride demonstrates significantly lower biofilm formation at 4, 24, 48 and 72 hours as compared to PEEK and titanium.
Proven Bacteriostatic Properties
The unique macrostructure, nano-texture, and elution kinetics of silicon nitride work together to prevent bacteria from adhering and effectively eliminate various strains, including S. epidermidis, S. aureus, MRSA, and even SARS-CoV-2.
Holistic Approach to Bacterial Deterrence
Silicon nitride's unparalleled combination of surface characteristics—topography, chemistry, pH, and wettability—creates a comprehensive and hostile environment for bacterial activity.
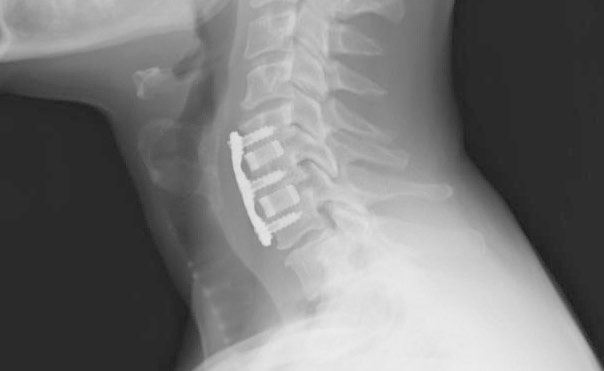
Enhanced Imaging
Silicon nitride implants provide favorable imaging across all modalities.
Clear Visibility
Silicon Nitride's lower density and semi-transparency allow X-ray beams to pass through with minimal scattering, resulting in sharp images of both the implant itself and the surrounding bone fusion in X-ray and CT scans.
Artifact-Free Imaging
The non-magnetic and bioinert properties of Silicon Nitride ensure accurate imaging of surrounding tissues without interference or adverse reactions within the body.
Reliable Results
The robust mechanical strength of Silicon Nitride ensures durability and resistance to deformation, providing consistent and accurate results throughout the manufacturing and imaging processes.
Experience the Future of Spine Care
Request a Demo of Our Silicon Nitride Implants
Discover firsthand the superior performance and benefits of Silicon Nitride implants. Schedule a demo today and see why leading surgeons are choosing our advanced solutions for their patients.


